Explain all chemical Test for alcohol and phenols. :max_bytes(150000):strip_icc()/ortho-meta-para-xylene-56a12db43df78cf772682c56.png)
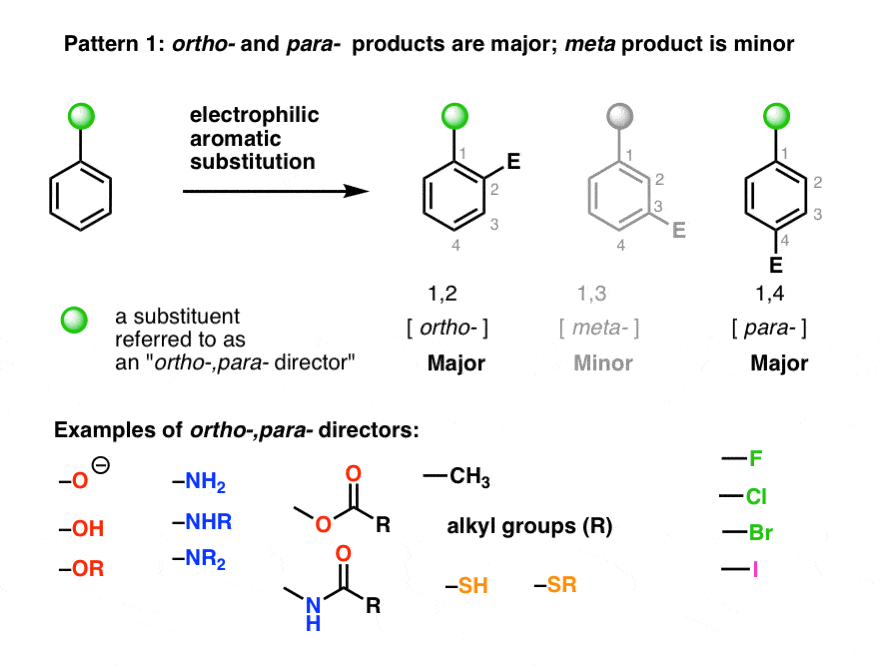
what are the list of tests for Aldehydes and Ketones.How shapes of molecules are related to the type of hybridization.How Ionic bond forms ? What are the properties.Toluene reacts considerably faster than benzene in all electrophilic substitutions. The methyl group is an activating group and is an ortho - para director. Several examples will illustrate more clearly what we mean by these terms.Īctivating Groups: Ortho - Para Directors Substituents in the second category tend to direct the incoming electrophile to the meta position. We call these groups ortho - para directors because they tend to direct the incoming group into the ortho and para positions. Substituents in one class tend to bring about electrophilic substitution primarily at the positions ortho and para to themselves. Those that cause the ring to be less reactive than benzene are called deactivating groups.We also find that we can divide substituent groups into two classes according to the way they influence the orientation of attack by the incoming electrophile. Those that cause the ring to be more reactive than benzene itself are called activating groups. We say, therefore, that substituent groups affect both reactivity and orientation in electrophilic aromatic substitutions.We can divide substituent groups into two classes according to their influence on the reactivity of the ring. When substituted benzenes undergo electrophilic attack, groups already on the ring affect both, the rate of the reaction and the site of attack. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
